 So far, reports of myocarditis after a booster dose are very rare, occurring in less than 1 in every 100,000 doses administered. A small number of myocarditis and pericarditis cases have been reported for booster doses. Reporting rates of adverse events following booster doses are very stable. In people who have recently had COVID-19, a 6-month interval is recommended before having their next scheduled dose. A booster dose is an additional vaccine dose given after the primary vaccine course. The TGA continues to monitor the safety of booster vaccine doses in adults. 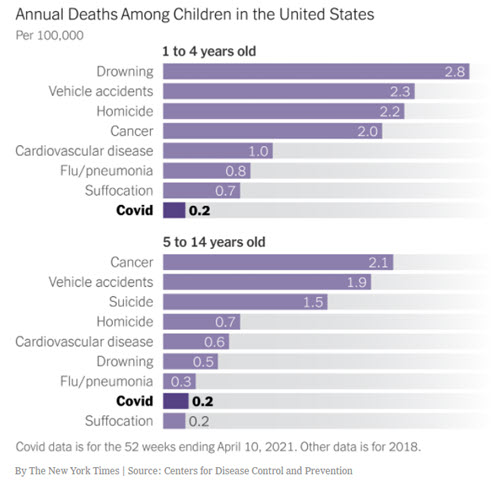
The most up-to-date vaccine recommendations for booster doses are available from ATAGI. More detail on vaccine safety in children and adolescents following vaccination is available in the safety report published on 15 December 2022. Reporting rates of adverse events following COVID-19 vaccination, including those for children and adolescents, are very stable. The TGA is closely monitoring adverse event reports in people aged under 18 years. The most up-to-date vaccine recommendations for children are available from ATAGI. Vaccine safety in children and adolescents These are recognised side effects of vaccination and are usually transient and mild. Skin reactions at the site of the injection are also common and can include pain, swelling, redness and an itchy rash. The most frequently reported side effects suspected to be associated with the vaccines include headache, muscle pain, fever, fatigue and nausea. Learn more about how to report a suspected side effect to a COVID-19 vaccine.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
